Simulation of Reflectance in Oral Tissue Using MCMatlab
Introduction
Clinical Motivation
Problem Statement
Background
Previous Work
Optical Tissue Properties in the Literature
| Tissue Layer | μa [cm⁻¹] 420 nm | μs [cm⁻¹] 420 nm | μa [cm⁻¹] 450 nm | μs [cm⁻¹] 450 nm |
|---|---|---|---|---|
| Superficial epithelium | 3.0 | 170 (keratinized) / 55 (non keratinized) | 2.1 | 140 (keratinized) / 40 (non keratinized) |
| Intermediate epithelium | 3.0 | 55 | 2.1 | 40 |
| Basal epithelium | 3.0 | 55 | 2.1 | 40 |
| Superficial stroma | 6.22 | 267 | 3.11 | 248.5 |
| Deep stroma | 6.22 | 267 | 3.11 | 248.5 |
| Blood | 262 | 37.8 | 30 | 31.1 |
Methods
Monte Carlo Simulation Principle
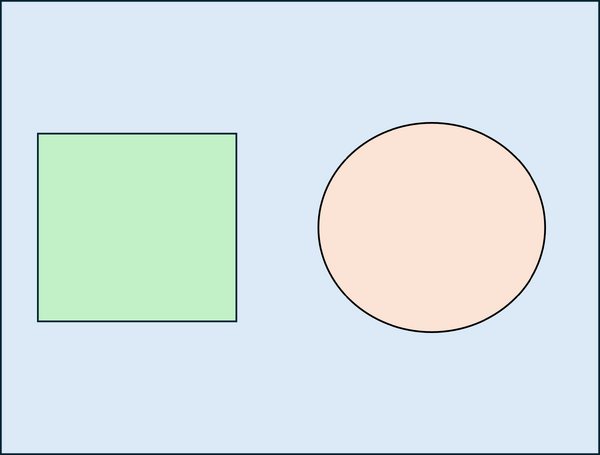
A simple Monte Carlo simulation can be used to estimate the value of π by comparing areas of a circle and a square. First, the geometric domain is defined, consisting of a square whose side is equal to twice the radius of the circle, so that the circle is fully inscribed within the square (see Figure 1a). A large number of points are then generated randomly and uniformly within the square. Each point is classified according to whether it lies inside the circle or outside, and the number of points falling within the circle is counted (see Figure 1b). The ratio of the number of points inside the circle to the total number of points in the square approaches the ratio of the areas of the circle and the square: . Multiplying this ratio by four therefore provides an estimate of π. This example illustrates how probabilistic sampling can be used to approximate geometric quantities.
|

|
| (a) Definition of the geometric domain | (b) Random point generation, classification, and counting |
| Figure 1: Basic Monte Carlo simulation to compute π | |
Monte Carlo Simulation for Light Propagation
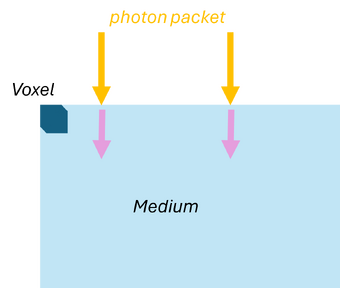
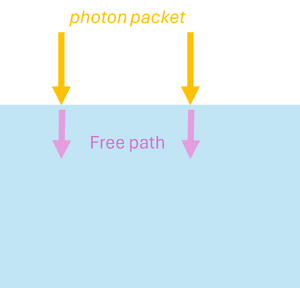
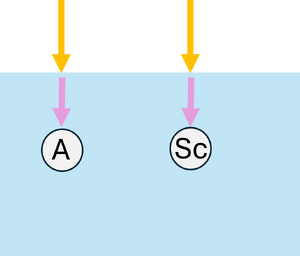
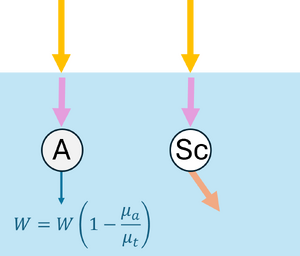
Monte Carlo simulations can also be applied to model the propagation of light in scattering and absorbing media, such as biological tissue. First, the geometric domain is defined, specifying the optical properties of the medium and the spatial grid where energy deposition will be recorded. A photon packet is then launched from the source with an initial weight (see Figure 2a). The photon propagates in a straight line over a free path drawn from an exponential distribution, , where (see Figure 2b). Upon reaching the end of the free path, an interaction occurs: the photon can be absorbed or scattered according to probabilities and . If scattering occurs, the anisotropy factor determines the preferential forward direction of the photon (see Figure 2c). During absorption events, the absorbed energy is and the photon weight is updated to , with the remaining weight continuing to propagate (see Figure 2d). By repeating these steps for many photon packets, the simulation statistically reconstructs macroscopic quantities such as fluence, reflectance, and transmittance in the medium.
|
|
|
|
| (a) Launching a photon packet | (b) Straight-line propagation over a free path | (c) Interaction event | (d) Updating the Photon Weight |
| Figure 2: Monte Carlo simulation for light | |||
Monte Carlo Matlab
Five Layer Model and Geometry
Results
Influence of Keratin on the Optical Response
Theoretical Results: Fluence
All graphs are presented in arbitrary units because the Monte Carlo simulations rely on photon weights W, which do not represent the exact number of photons. Fluence is calculated as , where wi is the weight of photon packet i, μa is the absorption coefficient of the medium traversed, and is the straight-line path length of the photon packet. In practice, fluence can be interpreted as the energy density within the tissue, or more intuitively, as a measure of how much light is present at a given depth z.

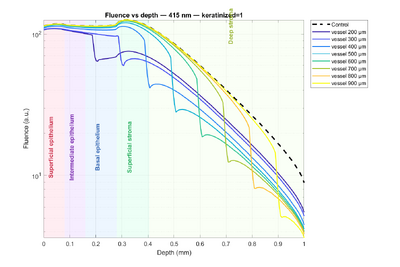
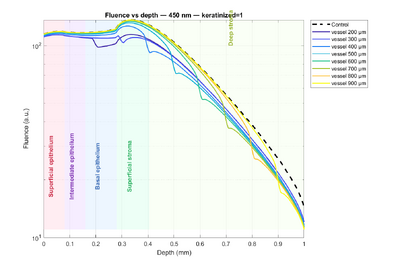
Let us first examine the impact of keratin on the tissue by referring to Figure 3 (dashed lines represent non-keratinized tissue, solid lines represent keratinized tissue). Two main observations can be made. First, in keratinized tissue, there is a noticeable fluence peak in the superficial epithelium. This is explained by the higher scattering compared to absorption: for example, at 415 nm, the scattering coefficients are 170 (keratinized) versus 55 (non-keratinized), and at 450 nm, 140 versus 40. As a result, light is more likely to be scattered than absorbed, leading to higher fluence. Second, starting from the intermediate epithelium, the trend reverses: fluence becomes slightly higher in non-keratinized tissue. This can be interpreted as light being partially “trapped” in the superficial epithelium of keratinized tissue due to strong scattering, reducing the amount of light reaching deeper layers.
Next, the influence of wavelength is considered. Comparing 415 nm and 450 nm, fluence is higher at 450 nm, which is consistent with lower absorption coefficients at this wavelength. Finally, a prominent feature appears between the epithelium and the stroma for all four curves: a large fluence increase. This arises because the absorption coefficient is effectively doubled in that region, which, according to the formula above, temporarily increases fluence. Beyond this peak, all curves gradually decrease with depth due to absorption.
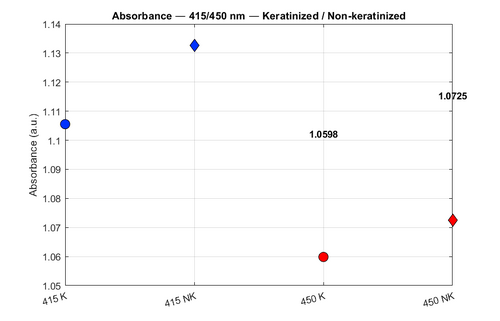
Measurable Results: Absorbance and Reflectance
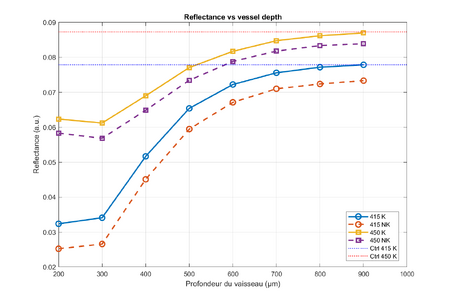
Influence of Blood Depth on the Optical Response
Theoretical Results: Fluence
Measurable Results: Absorbance and Reflectance
Discussion and Conclusions
Appendix
Our code can be found at (Github link).