Simulation of Optical Response in Oral Tissue
Introduction
Oral cancer remains a significant global health challenge, where early detection is the most critical factor in improving survival rates. Traditionally, screening has relied on standard white light examinations; however, this method is limited by the human eye's error-prone ability to detect subtle lesions that may not clearly manifest under typical examinations. To address these limitations, the VELscope has emerged as a promising, non-invasive, and rapipd technology for efficiently screening oral tissue.
Leading this technological shift is the VELscope, a handheld device that utilizes blue excitation light to visualize tissue abnormalities. Under VELscope illumination, healthy tissue emits a pale green fluorescence due to fluorophores such as collagen, whereas cancerous tissue typically appears as a distinct "dark spot" due to a loss of fluorescence. This contrast enhancement addresses the limitations of incandescent white light, clearly illuminating potential lesions that might otherwise go unnoticed.
Despite the clinical utility of the VELscope, a significant gap remains in the interpretation of these "dark spots." While the device is effective at identifying areas of low fluorescence, it lacks specificity regarding the biological cause of the signal loss; it could simply be a concentrated blood spot. Crucially, limited research has been conducted to distinguish between dark spots caused by cancer, and those caused by benign "blood spots" resulting from trauma, infection, or inflammation. Hemoglobin is a potent absorber of light in the blue-green spectrum, and therefore increased blood volume can mimic the optical appearance of a cancerous lesion by absorbing the fluorescence before it reaches the detector. This ambiguity presents a diagnostic challenge: a "dark spot" could represent a malignant breakdown of collagen, or it could simply represent a localized accumulation of blood due to another health issue. To improve the specificity of autofluorescence screening, it is essential to decouple the optical effects of blood absorption from true tissue fluorescence.
In this paper, we address this challenge by simulating the optical response of oral tissue using Monte Carlo modeling in MCmatlab. By computationally modeling the interaction of light with varying depths of blood vessels and tissue layers -- specifically comparing the response at the highly absorbing 415 nm wavelength versus the more penetrating 450 nm wavelength -- we aim to investigate the impact of blood on tissue reflectance. This study seeks to provide a theoretical framework for detecting benign blood absorption in oral tissue, potentially reducing false positives in optical oral cancer screening.
Background
Previous Work
The main objective of this work is to investigate how the presence of blood at different depths within oral tissue influences the optical response when illuminated at two wavelengths: 415 nm (near the peak of blood absorption) and 450 nm. Our analysis focuses on three experimentally measurable quantities and how they vary when a subsurface blood layer is present at different depths.
Several studies have explored how tissue morphology and site-specific optical properties affect measured signals. In particular, Pavlova et al. used Monte Carlo simulations to predict fluorescence spectra from normal, inflammatory and neoplastic oral sites, demonstrating the importance of layered tissue models and of using realistic, site-specific optical parameters. Their findings show that changes in scattering, absorption and layer thickness can substantially modify the detected optical signals.
Building on this framework, our approach adopts and adapts Pavlova’s five-layer oral tissue model for reflectance/fluence/absorbance simulations. We performed all photon-transport simulations with MCmatlab, a 3D voxel-based Monte Carlo tool that is well suited to implement layered geometries and to record depth-resolved energy deposition . For blood optical parameters we relied on the review by Jacques, while tissue parameters near 415 nm were taken from Pavlova’s dataset and interpolated as needed to obtain values at 415 nm and 450 nm.
Beyond Pavlova’s study, a number of other works have applied Monte Carlo methods to investigate the optical response of oral and skin tissues; these studies typically emphasize how variations in chromophore concentration (e.g., hemoglobin), scattering anisotropy, and layer thickness influence reflectance, transmittance and fluorescence. Together, the literature supports the two guiding principles of our study: (1) blood located at different depths produces distinctive signatures in depth-resolved optical signals, and (2) the sensitivity of those signatures depends strongly on illumination wavelength and on the local scattering/absorption properties of each layer.
In summary, our work follows an established Monte Carlo modeling tradition , while focusing specifically on the depth-dependent optical effect of subsurface blood layers at two diagnostic wavelengths (415 nm and 450 nm). The combination of a five-layer model, MCmatlab simulations and parameter sets from Jacques and Pavlova provides a coherent and reproducible basis for the simulations presented below.
Optical Tissue Properties in the Literature
| Tissue Layer | μa [cm⁻¹] 415 nm | μs [cm⁻¹] 415 nm | μa [cm⁻¹] 450 nm | μs [cm⁻¹] 450 nm |
|---|---|---|---|---|
| Superficial epithelium | 3.0 | 170 (keratinized) / 55 (non keratinized) | 2.1 | 140 (keratinized) / 40 (non keratinized) |
| Intermediate epithelium | 3.0 | 55 | 2.1 | 40 |
| Basal epithelium | 3.0 | 55 | 2.1 | 40 |
| Superficial stroma | 6.22 | 267 | 3.11 | 248.5 |
| Deep stroma | 6.22 | 267 | 3.11 | 248.5 |
| Blood | 262 | 37.8 | 30 | 31.1 |
Precise why keratin? Cheek/tongue
Methods
Monte Carlo Simulation Principle
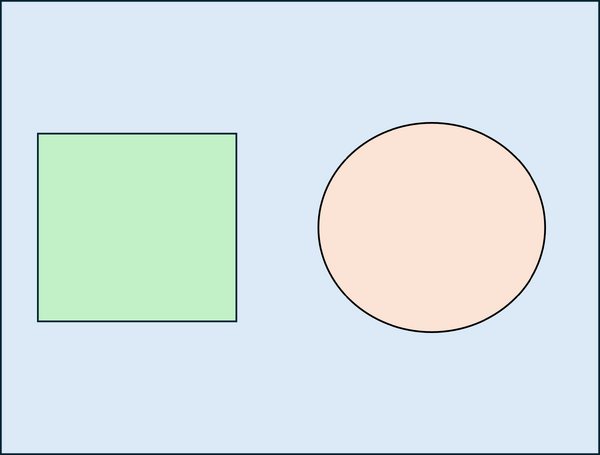
A simple Monte Carlo simulation can be used to estimate the value of π by comparing areas of a circle and a square. First, the geometric domain is defined, consisting of a square whose side is equal to twice the radius of the circle, so that the circle is fully inscribed within the square (see Figure 1a). A large number of points are then generated randomly and uniformly within the square. Each point is classified according to whether it lies inside the circle or outside, and the number of points falling within the circle is counted (see Figure 1b). The ratio of the number of points inside the circle to the total number of points in the square approaches the ratio of the areas of the circle and the square: . Multiplying this ratio by four therefore provides an estimate of π. This example illustrates how probabilistic sampling can be used to approximate geometric quantities.
The name “Monte Carlo” comes from the famous casinos in Monte Carlo, reflecting the stochastic (random) nature of the method. Monte Carlo simulations rely on the law of large numbers, meaning that in order to obtain accurate results, it is crucial to generate a sufficiently large number of random points so that the results converge to an asymptotic value.
|

|
| (a) Definition of the geometric domain | (b) Random point generation, classification, and counting |
| Figure 1: Basic Monte Carlo simulation to compute π | |
Monte Carlo Simulation for Light Propagation
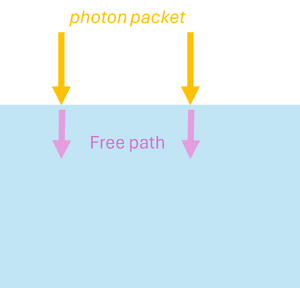
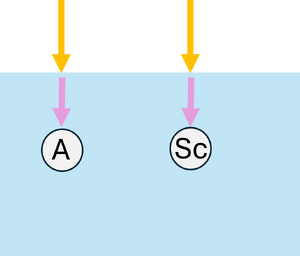
Monte Carlo simulations can be applied to model the propagation of light in scattering and absorbing media, such as biological tissue. First, the geometric domain is defined, specifying the optical properties of the medium and the spatial grid where energy deposition will be recorded. A photon packet is then launched from the source with an initial weight (see Figure 2a). The photon propagates in a straight line over a free path. The probability of an interaction occurring in the infinitesimal path interval is , where (see Figure 2b). Note that the free path follows an exponential distribution.
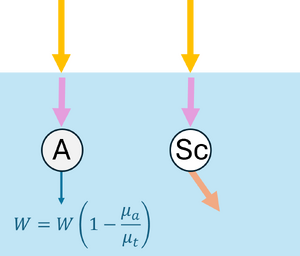
Upon reaching an interaction, the photon can be absorbed or scattered according to probabilities and . If scattering occurs, the anisotropy factor determines the preferential direction: corresponds to forward scattering, and larger values imply scattering at smaller angles, meaning the photon is deflected less from its initial direction. Here, , so scattering occurs mostly in the forward direction with small angles (see Figure 2c). During absorption events, the absorbed energy is , and the photon weight is updated to , with the remaining weight continuing to propagate (see Figure 2d).
By repeating these steps for many photon packets, the simulation statistically reconstructs macroscopic quantities such as fluence, reflectance, and transmittance in the medium.
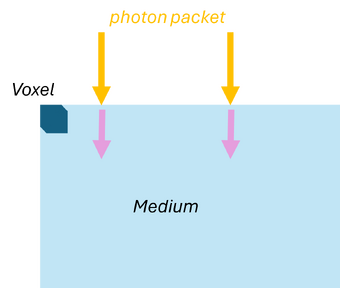
|
|
|
|
| (a) Launching a photon packet | (b) Straight-line propagation over a free path | (c) Interaction event | (d) Updating the Photon Weight |
| Figure 2: Monte Carlo simulation for light | |||
Monte Carlo Matlab
Five Layer Model and Geometry
Results
Influence of Keratin on the Optical Response
Theoretical Results: Fluence
All graphs are presented in arbitrary units because the Monte Carlo simulations rely on photon weights W, which do not represent the exact number of photons. Fluence is calculated as , where wi is the weight of photon packet i, μa is the absorption coefficient of the medium traversed, and is the straight-line path length of the photon packet. In practice, fluence can be interpreted as the energy density within the tissue, or more intuitively, as a measure of how much light is present at a given depth z.

Let us first examine the impact of keratin on the tissue by referring to Figure 3 (dashed lines represent non-keratinized tissue, solid lines represent keratinized tissue). Two main observations can be made. First, in keratinized tissue, there is a noticeable fluence peak in the superficial epithelium. This is explained by the higher scattering compared to absorption: for example, at 415 nm, the scattering coefficients are 170 (keratinized) versus 55 (non-keratinized), and at 450 nm, 140 versus 40. As a result, light is more likely to be scattered than absorbed, leading to higher fluence. Second, starting from the intermediate epithelium, the trend reverses: fluence becomes slightly higher in non-keratinized tissue. This can be interpreted as light being partially “trapped” in the superficial epithelium of keratinized tissue due to strong scattering, reducing the amount of light reaching deeper layers.
Next, the influence of wavelength is considered. Comparing 415 nm and 450 nm, fluence is higher at 450 nm, which is consistent with lower absorption coefficients at this wavelength. Finally, a prominent feature appears between the epithelium and the stroma for all four curves: a large fluence increase. This arises because the absorption coefficient is effectively doubled in that region, which, according to the formula above, temporarily increases fluence. Beyond this peak, all curves gradually decrease with depth due to absorption.
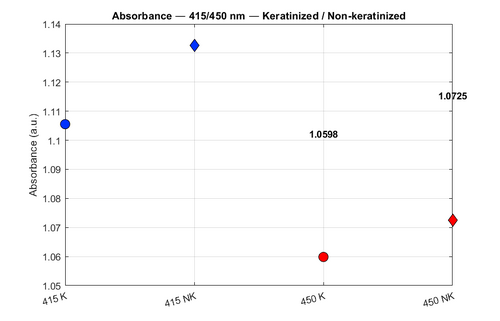
Measurable Results: Absorbance and Reflectance
Influence of Blood Depth on the Optical Response
Theoretical Results: Fluence
As a quick reminder, fluence is computed as , where wi is the weight of photon packet i, μa is the absorption coefficient of the medium traversed, and is the straight-line path length travelled by that photon packet. In practice, fluence represents the energy density within the tissue: intuitively, it measures how much light is present at a given depth z.
Let us first consider the 415 nm results (Figure 6a–b). The most striking feature across all curves is the abrupt drop in fluence at the location of the blood layer (the “blood carpet”). This sharp decrease is a direct consequence of the high absorption of blood at 415 nm: many photon packets are absorbed in the blood layer, so the local fluence falls precipitously. This characteristic behavior—an abrupt fluence reduction at a specific depth—suggests a robust way to detect the presence of blood in the tissue: a marked dip in the measured fluence profile at the corresponding depth is a clear indicator of a highly absorbing blood layer (see Figure 6a and 6b).
Immediately after the drop, the fluence shows a modest recovery (a small bump). This can be explained using the fluence formula: photon weights wi can only decrease with depth (we do not generate photons), so the only way for to increase locally is for the path lengths in the subsequent medium to increase. In other words, photons that survive the blood layer tend to travel longer straight-line distances in the less-absorbing tissue below, increasing the contribution of to the fluence and producing the observed small rise.
Comparing keratinized and non-keratinized tissues (Figure 6a vs 6b and 6c vs 6d), both exhibit the same dramatic drop when the blood layer is reached. The principal difference is the small superficial bump in keratinized tissue: as discussed earlier, the high scattering in the keratinized superficial epithelium redirects and retains light near the surface, producing a local fluence enhancement (the bump) that is absent or much smaller in non-keratinized tissue.
The same sequence of features appears at 450 nm (Figure 6c–d), but overall fluence values are higher than at 415 nm. This is expected because absorption coefficients are lower at 450 nm, so fewer photons are lost to absorption and more light penetrates deeper. Consequently, the fluence drop at the blood layer is less pronounced at 450 nm than at 415 nm: 415 nm lies closer to a blood absorption peak and is therefore more sensitive to the presence of blood, while 450 nm is comparatively less absorbed and shows a milder dip.
In summary, the combination of (i) a sharp fluence decrease at the blood depth, (ii) a small post-drop increase driven by longer photon path lengths in the less-absorbing tissue, and (iii) the superficial bump in keratinized tissue (due to strong scattering) provides a consistent, physically intuitive picture across wavelengths and tissue types (see Figure 6a–d).
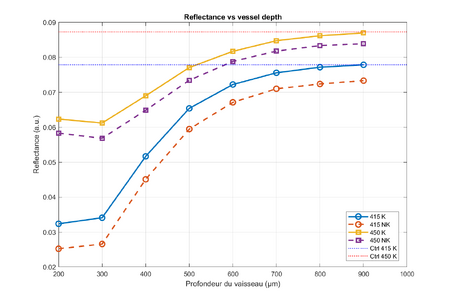
Measurable Results: Absorbance and Reflectance
Discussion and Conclusions
References
- Vibhute, N. A., Jagtap, S. V., & Patil, S. V. (2021). Velscope guided oral cancer screening: A ray of hope in early oral cancer diagnosis. Journal of Oral and Maxillofacial Pathology, 25(3), 548–549. DOI
- Pavlova, I. Fluorescence spectroscopy of oral tissue: Monte Carlo modeling with site-specific tissue properties.
- Sharma, R., Joshi, P., & Deopujari, A. Blood oxygen modelling from skin reflectance. Project report.
- Jacques, S. L. (2013). Optical properties of biological tissues: A review. Physics in Medicine & Biology, 58(11), R37–R61.
- MathWorks. (2023). MCmatlab: A Monte Carlo simulation for photon transport in 3D voxel space. Link
- Radiative Transfer Equation. ScienceDirect Topics. Link
- Salomatina, E. et al. (2006). Optical properties of normal and cancerous human skin in the visible and near-infrared spectral range. Journal of Biomedical Optics, 11(6), 064026.
- Morgan, D. (2024). How does the VELscope work? VELscope. Link
Appendix
Our code can be found at (Github link).