Imaging Strategies for Enhancing Blood Visibility in Oral Tissue
Introduction
Early detection is critical for improving outcomes in oral cancer, motivating a range of noninvasive optical approaches that aim to flag suspicious tissue changes at the point of care. Among these, fluorescence-based imaging is attractive because collagen can emit in the green band when excited with blue/violet light. However, interpreting oral fluorescence images remains challenging because a decrease in measured fluorescence is not specific: reduced signal may arise from malignant change, but it can also reflect benign physiological variation—most notably blood content. In the oral cavity, this ambiguity is especially pronounced because hemoglobin strongly absorbs around 415 nm, so vascularized regions can appear “dark” even in otherwise healthy tissue.
Alongside this physiological confound are the optics of the measurement itself. Oral tissue is humid and highly reflective, producing strong specular highlights and additional scattering that reduce contrast and can mask the very absorption differences needed to localize blood. Spectrally, the visible range is particularly problematic because hemoglobin absorption overlaps both excitation/reflectance pathways and tissue autofluorescence, whereas the near-infrared (∼700-900 nm) reduces hemoglobin absorption and autofluorescence background while improving penetration. These challenges motivate imaging strategies that suppress surface reflections and better isolate signals attributable to hemoglobin versus underlying tissue structure.
In this study, we investigate practical imaging strategies for enhancing blood visibility in oral tissue by comparing:
- cross-polarized reflectance imaging to suppress specularity and glare
- fluorescence imaging with spectral filtering to isolate emission and increase vascular contrast
By evaluating these approaches in a controlled acquisition setup and analyzing the channels most informative for reflected versus emitted light, we aim to clarify the tradeoffs of each method and inform future systems that more robustly separate blood-driven absorption from tissue changes relevant to diagnosis.
Background
Optical imaging techniques have been increasingly explored for noninvasive evaluation of oral tissue properties and early detection of pathological changes. One fundamental challenge in optical imaging of the oral cavity is the strong absorption of visible light by hemoglobin (Hb), which significantly affects both reflectance and fluorescence signals captured from tissue (Sweeny et al., 2011). Hemoglobin absorbs light strongly in the visible spectrum, particularly near 415 nm, and this property underlies much of the contrast observed in reflectance imaging modalities used to detect vascular features (Sweeny et al., 2011).
Reflectance imaging has been investigated clinically as an adjunct to conventional examination for screening oral lesions. Reflectance spectra of oral mucosa reveal absorption features attributable to oxygenated hemoglobin that correlate with increased microvasculature in neoplastic tissue (Tiwari et al., 2019; Sweeny et al., 2011). Optical devices using specific illumination bands, such as green‑amber light (~540–575 nm), aim to enhance visualization of underlying vasculature by exploiting these absorption characteristics (Tiwari et al., 2019).
Endogenous tissue fluorescence provides an additional source of contrast in optical imaging. Fluorescence arises when endogenous fluorophores absorb excitation light and re‑emit it at longer wavelengths due to the Stokes shift, enabling emission signals to be separated from reflected light (Wu et al., 2018). In oral tissue, structural proteins such as collagen contribute significantly to autofluorescence, and changes in these signals have been associated with pathological transformation (Pavlova et al., 2008; Tiwari et al., 2019). Studies show that dysplastic and malignant tissues can exhibit decreased fluorescence relative to normal tissue, partly due to alterations in fluorophore composition and increased absorption by blood and other chromophores (Pavlova et al., 2008; Tiwari et al., 2019).
Polarization imaging offers a method to reduce specular reflection from tissue surfaces by filtering light based on its polarization state. While polarization techniques have been used in various biomedical applications to probe tissue structure and reduce surface reflectance (Tuchin, 2016), their application in oral tissue imaging remains relatively exploratory. Polarization‑based imaging can improve contrast by suppressing surface reflections that otherwise mask subsurface optical properties, although most existing oral applications focus on polarization‑sensitive optical coherence tomography rather than wide‑field polarization reflectance imaging (Yoon et al., 2015).
Handheld and wide‑field imaging systems that combine reflectance and fluorescence have been evaluated clinically, with mixed results regarding sensitivity and specificity for oral cancer screening (Sweeny et al., 2011; Tiwari et al., 2019). These findings support continued investigation into systematic imaging strategies that isolate absorption and emission effects while minimizing confounding surface reflection, particularly under controlled illumination conditions.
Methods
We compared two imaging configurations designed to increase the visibility of blood-driven absorption in oral tissue: (1) cross-polarized reflectance imaging to suppress surface glare, and (2) fluorescence imaging with spectral filtering to isolate emitted light. Both setups used a DSLR camera (Nikon D500) and a fixed geometry in which the camera and illumination source were placed at approximately 45° relative to the target (lower lip) or a Macbeth Color Checker (MCC) used for reference and setup validation.


Cross polarization methodology
Cross-polarization was implemented with two polarizers: one mounted on the illumination source and one mounted on the camera lens. Images were acquired in parallel orientation, which allows specular reflections to reach the sensor, and crossed orientation, which suppresses specular reflection to better reveal subsurface reflectance and absorption patterns.
The camera (Nikon D500) and light source were positioned at ~45° relative to the lips or MCC, with the subject centered along the illumination and imaging axes; this geometry intentionally permits specular reflections to occur so that the effect of cross-polarization on suppressing glare can be observed. One polarizer on the light source was kept fixed, while the polarizer on the camera lens was rotated to capture two images (parallel vs. crossed).
Is cross-polarization using circular polarizers less effective than cross-polarization using linear polarizers? With circular polarizers, the polarization change happens more gradually through tissue; less of the returned signal actually switches from one handedness to the other; and more good signal is rejected along with the bad signal.
What if you only use a polarizing filter on the camera? If unpolarized light (regular light) is shined at the tissue and only put a polarizing filter on the camera, a one-sided measurement is being done. The camera only sees light with one specific polarization orientation. This means most of the signal is lost because a polarizer blocks roughly 50% of unpolarized light; surface reflections and scattered light still interfere with deeper tissue imaging; and the light is not being filtered and rejected in a controlled way.
Linear polarization was treated as preferable for this application because it provides sharper differentiation between blocked surface glare and transmitted subsurface scatter than circular polarization.
Fluorescence imaging methodology
Fluorescence imaging used ~415 nm excitation to elicit green-band emission (approximately 500–600 nm) from oral tissue (e.g., collagen fibers). Blood appears dark because hemoglobin absorbs both the excitation and portions of the emitted light (including around 540 and 580 nm), increasing vessel contrast against brighter fluorescing tissue.
A 415 nm short-pass filter was placed on the light source to ensure excitation wavelengths below 415 nm reached the tissue. A 475 nm long-pass filter was placed on the camera lens to block reflected excitation light while passing emitted fluorescence in the 500–600 nm range.
As in cross-polarization, the camera and blue LED were positioned at ~45° relative to the lips or MCC with the subject centered on the illumination/imaging axes. Images were then acquired for analysis focused on the fluorescence band.
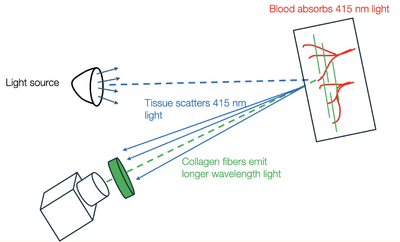
Image formats and preprocessing
To examine the impact of camera processing, both “unprocessed” (raw-like, maximum post-processing flexibility) and “processed” (camera-applied settings such as white balance/sharpening and compression) imagery were compared.
Use of MatLab
All quantitative viewing/inspection was performed on the unprocessed raw sensor images in MATLAB using a custom script, s_crossPolarizationVsFluorescence.m, which reads Bayer-patterned sensor data, parses image metadata including the CFA pattern, and outputs either an RGB rendering or a single color channel for analysis.

Results
Images from the Lower Lip Experiments
Conclusions
References
Pavlova, I., Williams, M., El-Naggar, A., Richards-Kortum, R., & Gillenwater, A. (2008). Understanding the Biological Basis of Autofluorescence Imaging for Oral Cancer Detection: High-Resolution Fluorescence Microscopy in Viable Tissue. Clinical Cancer Research : An Official Journal of the American Association for Cancer Research, 14(8), 2396–2404. https://doi.org/10.1158/1078-0432.CCR-07-1609
Sweeny, L., Dean, N. R., Magnuson, J. S., Carroll, W. R., Clemons, L., & Rosenthal, E. L. (2011). Assessment of Tissue Autofluorescence and Reflectance for Oral Cavity Cancer Screening. Otolaryngology, 145(6), 956–960. https://doi.org/10.1177/0194599811416773
Tiwari, L., Kujan, O., & Farah, C. S. (2020). Optical fluorescence imaging in oral cancer and potentially malignant disorders: A systematic review. Oral Diseases, 26(3), 491–510. https://doi.org/10.1111/odi.13071
Tuchin, V. V. (2016). Polarized light interaction with tissues. Journal of Biomedical Optics, 21(7), 071114. https://doi.org/10.1117/1.jbo.21.7.071114
Wu, C., Gleysteen, J., Teraphongphom, N. T., Li, Y., & Rosenthal, E. (2018). In-vivo optical imaging in head and neck oncology: basic principles, clinical applications and future directions. International Journal of Oral Science, 10(2), 10. https://doi.org/10.1038/s41368-018-0011-4
Yoon, Y., Won Hyuk Jang, Xiao, P., Kim, B., Wang, T., Li, Q., Ji Youl Lee, Chung, E., & Ki Hean Kim. (2015). In vivo wide-field reflectance/fluorescence imaging and polarization-sensitive optical coherence tomography of human oral cavity with a forward-viewing probe. Biomedical Optics Express, 6(2), 524–524. https://doi.org/10.1364/boe.6.000524