Lalwani Balasingam Olazarra Saffari: Difference between revisions
imported>Student221 |
imported>Student221 |
||
| Line 60: | Line 60: | ||
These are the absorption and emission spectra we recorded for NADH at various wavelengths. | These are the absorption and emission spectra we recorded for NADH at various wavelengths. | ||
[[File:nadh.png| | [[File:nadh.png|800px]] | ||
<div align="center">'''Figure 4''': The leftmost plot illustrates the absorption spectrum of NADH at an emission of 415 nm. The following two plots (in the middle and the right) are emission spectra where NADH is excited at 293 nm and 317 nm, respectively. Note that, on the rightmost emission spectrum, the concentration of NADH was doubled before being excited at 317 nm.</div> | <div align="center">'''Figure 4''': The leftmost plot illustrates the absorption spectrum of NADH at an emission of 415 nm. The following two plots (in the middle and the right) are emission spectra where NADH is excited at 293 nm and 317 nm, respectively. Note that, on the rightmost emission spectrum, the concentration of NADH was doubled before being excited at 317 nm.</div> | ||
Revision as of 21:58, 12 December 2019
Introduction
This quarter, our team worked on characterizing fluorophores purchased by the Valdez Lab in the Department of Otolaryngology, Head and Neck Surgery at the Stanford School of Medicine to determine optimal materials for cholesteatoma visualization. The fluorophores we used in our measurements include elastin, keratin, NADH, and collagen. Our inspiration to pursue a cholesteatoma imaging project was derived from work being done in the Valdez lab as well as oral imaging work being done by Dr. Joyce E. Farrell and Zheng Lyu in the Department of Electrical Engineering. We hope our work can shed some light on which fluorophores may be best for producing a label-free method in congenital cholesteatoma detection via autofluorescence microscopy. To aid these efforts, our teams worked on characterizing and analyzing such fluorophores to determine the optimal selections for clinical use given their respective absorption and emission spectra. Following these measurements, we designed a fluorophore matrix that accommodated the fluorophores we identified to enable further measurements and characterization to be performed using the imaging mechanisms developed by Dr. Farrell and Zheng Lyu. While our group focused on using this matrix for measurements of our chosen fluorophores, the matrix was designed to enable users to customize the matrix by inserting and removing their own selected fluorophores. While our project allowed us to gain a great deal of information about the fluorophores and expand upon our own understanding of how light interacts with materials, we plan on refining our methods and taking further measurements in the new year to come.
Background
In this section, we will provide both the clinical and technical background that inspired our motivation to pursue this project. Additionally, we will discuss the reasons we deemed fluorescence to be an ideal diagnostic tool given our clinical objective.
Clinical Background
We were driven to find a label-free method for autofluorescence imaging of cholesteatomas. Cholesteatomas are non-malignant, non-cancerous growths that typically form behind the tympanic membrane [1, 2]. It is similar to a cyst, and often occurs in people who are prone to otitis media (or middle ear infections), and is often congenital [2, 3]. Our objective is to create a label-free method to characterize biomolecular changes in the keratinized epithelial tissue by exploiting the molecular specificity of autofluorescence spectroscopy. Specifically, we will measure the spectral signatures of biological molecules like NADH and elastin, both of which are naturally found in the body and can fluoresce upon absorption of ultraviolet (UV) or visible light. Ultimately, being able to quickly and reliably visualize these cholesteatomas is desirable for early diagnosis and complete resection of the lesions, in order to better preserve the patient’s hearing ability at the time of surgery.
Technical Background
To better visualize neoplastic regions, researchers have focused on creating optical imaging methods that exploit various interactions between light and tissue [4]. For our purposes, we are interested in the autofluorescence of tissue behind the tympanic membrane. Autofluorescence is a product of the numerous fluorophores in the middle ear cavity (such as collagen and elastin), and is particularly sensitive to changes in tissue morphology and biochemical alterations of neoplasia [5, 6]. Therefore, tissue autofluorescence is being harnessed as a diagnostic tool for oral cancers, various head and neck lesions, and for our purposes, in cholesteatomas. By exciting fluorophores in the ear with UV or even visible light, the emitted light will fluoresce at longer wavelengths. We can measure this emission spectra following excitation and, with the use of an absorbing filter to block reflected illumination, can visualize the fluorescence with our eyes [4]. For our purposes, we will not be visualizing these fluorophores with our eyes so much as we will be using a fluorometer to accurately measure the emission spectra and record the peak wavelength following excitation.
Methods
We acquired four fluorophores (NADH, collagen, elastin, and keratin), then measured the fluorescence signal produced by each of them using both a fluorometer and the OralEye camera. The purpose of our initial fluorescence measurements, which were performed using a fluorometer, was to determine whether we were able to measure emission spectra consistent with literature values for each of our fluorophores. Secondary measurements, which were performed using the OralEye camera, sought to determine whether we were able to effectively measure similar emission spectra using the 385 nm excitation light source associated with the OralEye camera.
Fluorometer Measurements with Haribo's Fluorolog Device
The fluorometer we used for the measurement of the fluorophores was made available to us courtesy of the Stanford Department of Chemistry in Stauffer 1. Figure 1 is an image of the machine that allowed us to measure the emission and absorption spectra of the fluorophores.

The fluorometer’s excitation wavelengths range from 250 nm to 400 nm, while the emission spectrum has a range between 250 nm to 1000 nm. Therefore, we felt that this tool would be particularly useful in our measurements as its dynamic range met the specifications required by our fluorophores.
As for the fluorophores, we tried to keep the concentrations consistent within each cuvette so that we could effectively compare spectra between NADH, elastin, collagen, and keratin. For our liquid samples (keratin and elastin when mixed with deionized (DI) water), we put ~30 ul into 1.5 mL of DI water in a clear cuvette. For NADH, which came in the form of small dip-n-dot like samples, we placed between 3-5 dots (depending on the size of the dots) in 1.5 mL of DI water in the cuvette. Finally, for collagen, which was in a cotton-ball-like form, we evenly divided the sample into 5 pieces and placed 1 piece in the cuvette for spectral measurements. Based on this concentration level, we set the fluorolog’s slit size to 3.0 mm in order to produce emission spectra that did not have disproportionately high intensities. As a reference, concentrated solutions are typically those that look cloudy in the cuvette; and those solutions that are more heavily concentrated must have a smaller slit width in order to appropriately measure the fluorescence at a reasonable intensity.
Measurements with the OralEye Camera
In order to determine whether we were able to measure fluorescence using the OralEye camera, we built a matrix that not only accommodated our water-soluble fluorophores, but also accommodated standard fluorescent microscope slides (and similarly-prepared, alternative fluorophores identified by the user). A three-dimensional rendering of the matrix, which was modeled after standard color calibration targets, is pictured in Figure 2. Performing measurements using the fluorescent calibration targets allowed us to determine whether we were able to measure any fluorescence at all, while performing similar measurements using our chosen fluorophores allowed us to attempt to measure and compare their respective fluorescence signals with the results obtained using the fluorometer.

Prior to performing the fluorophore fluorescence measurements, we prepared four standard glass microscope slides: one for each of the respective fluorophore solutions. Sample preparation varied depending on the state in which the fluorophores were provided, but was largely consistent with the sample preparation described in the fluorometer measurements section. For keratin and elastin, which were provided in liquid form, we dissolved approximately 20uL of the fluorophore in 1mL of DI water. For NADH and collagen, the concentrations were slightly less straightforward. The NADH provided took the form of a collection of non-uniform beads; to prepare this sample, we dissolved approximately five of the beads in 20uL of distilled water. Finally, the collagen was presented as a cotton-ball-like substance; to prepare this sample, we tore off a small portion of the collagen substance, then dissolved it in 20uL of distilled water. After the fluorophore solutions were prepared, we sealed them by placing glass cover slides on top.

Our four fluorophore samples were then installed in the aforementioned matrix in order to begin performing fluorescence measurements using the OralEye camera. We fixed the matrix to an optical stand, which was then fixed to an optical table. The optical stand allowed us to move the matrix vertically in order to capture data corresponding to both the top and bottom row samples. The OralEye camera was installed on a track that allowed us to move the camera/light source horizontally in order to illuminate and capture data corresponding to both the left and right column samples. In order to actually capture the emission spectra, we situated a spectroradiometer approximately one meter from the matrix. Because the spectroradiometer, alone, does not have a dynamic range high enough to distinguish a weak emitted fluorescence signal from a strong reflected light signal, we equipped the spectroradiometer with a Y44 longpass filter. By installing this filter, we were able to filter out a large fraction of the signal produced due to light reflected from the 385 nm light source. (Further discussion regarding the efficacy of this filter with respect to our particular results can be found in the “Results” and “Conclusions” sections.)
Results
Results from Fluorometer Measurements
We found the following spectra for the NADH, collagen, elastin, and keratin fluorophores using Haribo’s fluorolog. Additionally, we chose to excite these fluorophores at specific wavelengths based on work being conducted in the Valdez lab and methods used in previous literature [4, 7, 8, 9].
NADH
These are the absorption and emission spectra we recorded for NADH at various wavelengths.
These results are consistent with our those recorded in previous literature [7]. Out of all the spectra recorded from fluorophores, NADH was the most consistent with previously recorded results.
Collagen
These are the absorption and emission spectra we recorded for collagen at various wavelengths.
<<INSERT PIC>>
These results are not consistent with results from previous literature, with the peaks shifted to the left of the spectrum [8]. The reason behind these readings being at a lower wavelength are discussed in greater detail in the troubleshooting section below.
Elastin
These are the absorption and emission spectra we recorded for elastin at various wavelengths.
<<INSERT PIC>>
These results are nearly, but not completely consistent with results from previous literature [8]. While the absorption spectra is relatively close to what has been seen in previous findings, the emission spectrum for elastin typically is more prominent at around 400 nm. The rightmost emission spectrum is closer to this value, but is just a hair shy. Additionally, these peaks are broader than typical spectra for elastin.
Keratin
These are the absorption and emission spectra we recorded for keratin at various wavelengths.
<<INSERT PIC>>
These results are nearly, but not completely consistent with results from previous literature [9]. While the absorption spectra is relatively close to what has been seen in previous findings at high keratin concentration, the emission spectrum for keratin typically is more prominent at ~390 nm. Additionally, we also see a secondary, smaller peak popping up around ~360 nm. We explain the reasons behind this error in more detail in the troubleshooting section.
Troubleshooting the Measurements
One of the main reasons why our spectra may differ from those recorded for these fluorophores in previous literature is due to cross-contamination of the fluorophores. Before beginning our experiment, we were prepared to measure our samples using cuvettes with beveled edges. This produced a great deal of scattering, and so we switched over to using clear cuvettes without any beveled edges. Unfortunately, we did not have enough of these clear cuvettes for each sample, so we had to reuse the cuvette for numerous samples. While we did try our best to clean and remove any remnants of previous fluorophores, we would be remiss to not recognize the potential for certain fluorophores being mixed with one another.
Another reason why some of the emission spectra may be shifted from those in previous literature may arise from us not exciting all of the fluorophores at ideal wavelengths. In particular, we are most interested in redoing these measurements by exciting the fluorophores closer to the visual spectrum at 385 nm.
While both of these potential sources of error could have skewed some of our measurements, they are relatively easy to fix by ordering a full panel of clear cuvettes and by adjusting our wavelength excitation on the fluorolog.
Results of OralEye Camera Measurements
In order to understand the data we collected for our fluorophore measurements, we plotted the emission spectra corresponding to each of our fluorophores as a function of wavelength. This plot can be seen in Figure 8. In inspecting this plot, we made two primary observations. First, we noted that all four of our fluorophores demonstrated a sharp increase in measured fluorescence at a wavelength of approximately 425 nm, peaking shortly thereafter. The fact that all four of our fluorophores peaked in this region was not necessarily worrisome of and by itself, as we expected very little signal below 425 nm due to the presence of the Y44 filter, which is ideally designed to only pass light corresponding to wavelengths greater than 425 nm. As such, it seemed reasonable that we would see a spike in all four of the signals at wavelengths beyond this threshold. The second observation we made was that the peak magnitude of the measured fluorescence for three of our four fluorophores was nearly identical (exact values for maxima are specified in the legend of Figure 8). This feature of the plot was slightly more concerning, as it suggested the possibility that we were measuring the same signal for all of our fluorophores despite the fact that we expected different emission spectra. This raised concerns regarding the integrity of our data, and pointed to the possibility that we measured reflected light rather than a true fluorescence signal.
In order to understand the data we collected for our fluorophore measurements, we plotted the emission spectra corresponding to each of our fluorophores as a function of wavelength. This plot can be seen in Figure 8. In inspecting this plot, we made two primary observations. First, we noted that all four of our fluorophores demonstrated a sharp increase in measured fluorescence at a wavelength of approximately 425 nm, peaking shortly thereafter. The fact that all four of our fluorophores peaked in this region was not necessarily worrisome of and by itself, as we expected very little signal below 425 nm due to the presence of the Y44 filter, which is ideally designed to only pass light corresponding to wavelengths greater than 425 nm. As such, it seemed reasonable that we would see a spike in all four of the signals at wavelengths beyond this threshold. The second observation we made was that the peak magnitude of the measured fluorescence for three of our four fluorophores was nearly identical (exact values for maxima are specified in the legend of Figure 8). This feature of the plot was slightly more concerning, as it suggested the possibility that we were measuring the same signal for all of our fluorophores despite the fact that we expected different emission spectra. This raised concerns regarding the integrity of our data, and pointed to the possibility that we measured reflected light rather than a true fluorescence signal.

In an effort to better understand our results, we plotted the emission spectra of our fluorophores alongside the filtered spectrum of the 385 nm light source (see Figure 9). That is, we multiplied the spectrum of the 385 nm light with the transmittance of the Y44 longpass filter, then compared the resulting spectrum with the emission spectra of our fluorophores. By plotting these together, we were able to assess how the magnitude of the filtered, reflected light compared with the magnitude of the measured fluorescence signal corresponding to each of our fluorophores. As Figure A.A indicates, even after the transmittance profile of the Y44 longpass filter was applied, some amount of the reflected 385 nm was transmitted. Furthermore, the amplitude of the reflected light signal was visibly larger than the amplitude of any of the fluorescence signals at 425 nm. This suggested to us that there was, in fact, a strong possibility that our fluorescence measurements were significantly distorted by light reflected from our light source.

In order to further explore the possibility that we had not measured a legitimate fluorescence signal for our fluorophores, we plotted the normalized fluorescence emission spectra for all four of our fluorophores and compared them with previously-established literature values. Figure 10 summarizes the emission spectra results obtained by Yang, et. al. while Figure 11 shows the normalized emission spectra for each of the four fluorophores we measured. While we would not expect the exact magnitude of our fluorescence measurements to match those obtained by Yang, et. al. due to different normalization criteria and excitation spectra, we would expect to see the same trend in terms of relative magnitudes of measured fluorescence at a given wavelength. For example, at a wavelength of 500 nm, we would expect to see (in order of lowest magnitude response to highest magnitude response): collagen, elastin, and NADH, with elastin and NADH showing significantly higher fluorescence signals than collagen. However, our results show (in the same increasing order): elastin and collagen (where elastin and collagen are nearly indistinguishable at 500 nm), then NADH. This discrepancy in our values versus previously established literature values further supported our hypothesis that we measured reflected light rather than fluorescence.


After establishing that we did not measure a robust fluorescence signal for our fluorophores, we wanted to determine whether or not we were able to effectively measure the stronger, more robust fluorescence signal associated with our fluorescent calibration slides. This would help us to clarify whether our undesirable fluorophore measurements were rooted in the weak fluorescence of those fluorophores, or a general inability to measure fluorescence using our previously-described experimental configuration and measurement schema. Just as we had done in our fluorophore measurement analysis, we began this process by plotting the emission spectra of the calibration slides alongside the filtered reflected light spectrum. From this initial plot, pictured in Figure 12, it immediately became clear that the fluorescence signal associated with the calibration slides was far more robust when compared with the signal associated with the reflected light than the fluorophores were. That is, the magnitude of the fluorescence signal for the calibration slides was much larger than the magnitude of the signal associated with the reflected light at the wavelengths we were interested in. This indicated that the signals we were measuring at or above 425 nm were likely due to actual fluorescence rather than reflected light. As such, we concluded that our experimental configuration could, in fact, measure fluorescence provided that the fluorescence produced by the sample was greater in magnitude than that of the reflected light.

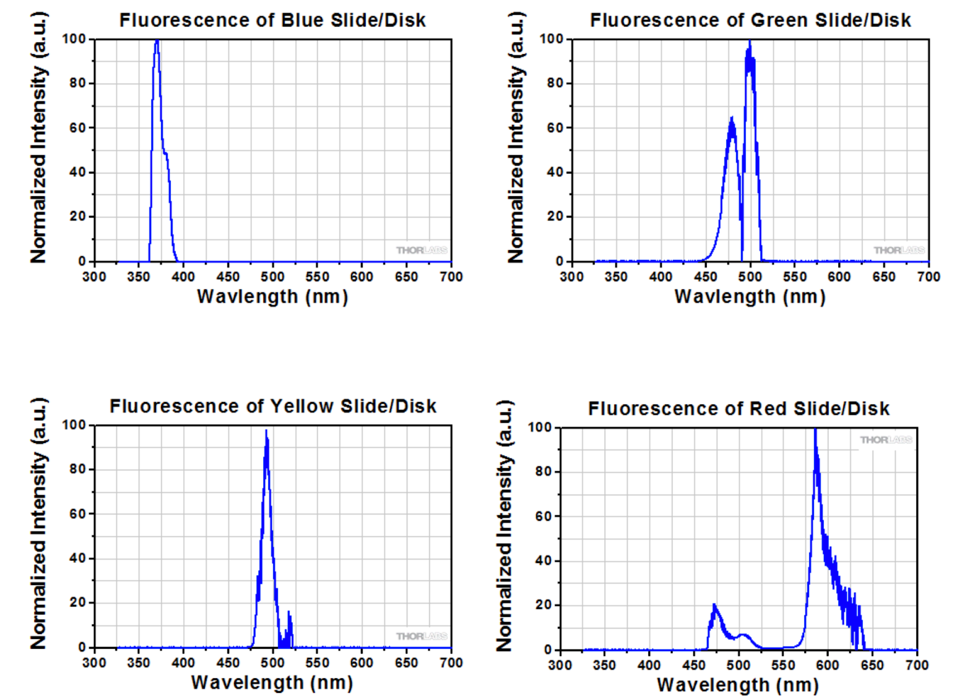
To further assess the integrity of the calibration slide measurements, we compared the normalized emission spectra with the emission spectra provided by the slide manufacturer. Figure 13 shows a plot of the normalized emission spectra of the fluorescent calibration slides after the Y44 filter transmittance profile was applied. When we compare this with the expected results published by the slide manufacturer, which are summarized in Figure 14, we saw fairly consistent results for all of the calibration slide emission spectra except for the results associated with the blue slide. While the manufacturer results indicate a blue peak at approximately 375 nm, our blue peak occurs at approximately 458 nm. While we are not entirely sure about why we saw this inconsistency, one reason might be that the expected blue emission peak occurs below 425 nm, which is the Y44 filtering threshold. As such, we believe that we may have been unintentionally filtering out a portion of the blue calibration slide’s emission spectrum, thus distorting the results.

.
Conclusions
Throughout our work on this project, particularly during the OralEye measurement analysis, we became acutely aware of the challenges associated with measuring weak fluorescence signals in the presence of even the smallest quantities of reflected light. Despite our efforts to diminish the effects of light reflected by our light source by implementing a Y44 longpass filter, we still saw sufficient enough transmission such that our fluorophore fluorescence signal was indistinguishable. While we regret that we were not able to measure a more robust fluorescence signal for our selected fluorophores, we feel encouraged by the promise we saw in our calibration slide measurements, and feel confident that meaningful fluorescence measurements can be collected if the issues of reflected light leakage are addressed going forward. For recommendations regarding next steps for this particular portion of the project, please see the “Future Work” section below.
Future Work
In the future, we plan to evaluate the effects of different concentrations on our ability to distinguish a fluorescence signal from the background reflected light signal. Our experiments with the fluorometer indicate that concentration can have a noticeable effect on our ability to measure fluorescence. Assuming a similar dependency on concentration in our OralEye camera set-up, we would like to perform many experiments to find the “sweet-spot” concentration value that gives us measurable fluorescence. By the same token, we would like to perform more measurements on the fluorometer by exciting our fluorophores at the 385 nm wavelength, to better compare the measurements between the OralEye Camera and the emission spectra. This will also help us find emission spectra that are consistent with those recorded in previous literature.
In order to acquire more meaningful data using the OralEye camera set-up, we must address the issue of measuring weak fluorescence in the presence of a relatively strong reflected light signal. One obvious way to do this is to implement a more effective filter in order to prevent excessive reflected light from “leaking” into our fluorescence measurements. One possibility would be to apply a filter with an even higher threshold, such as a Y48 or Y50 filter (with cut-off wavelengths of ~480 nm and ~500 nm, respectively), to further reduce the impact of the light reflected from the 325 nm light source. As mentioned earlier, another possibility would be to increase or decrease the concentration of the fluorophore samples we measure; doing so might increase the signal produced by the fluorophores, and thus reduce the issues associated with the weak signal we saw in our earlier measurements. By exploring these sorts of solutions, we feel confident that a robust set of measurements can be acquired for our selected fluorophores.
References
[1] E. L. Derlacki and J. D. Clemis. “Congenital cholesteatoma of the middle ear and mastoid.” Ann. Otol. Rhinol. Laryngol. 1965, 74, 706.
[2] M. J. Levenson, S. C. Parisier, P. Chute, S. Wenig and C. Juarbe. “ A review of twenty congenital cholesteatomas of the middle ear in children.” Otolaryngol. Head. Neck. Surg. 1986, 94, 560.
[3] G. T. Richter and K. H. Lee. “Contemporary assessment and management of congenital cholesteatoma.” Curr. Opin. Otolaryngol. Head. Neck. Surg. 2009, 17, 339.
[4] Shin D, Vigneswaran N, Gillenwater A, Richards-Kortum R. “Advances in fluorescence imaging techniques to detect oral cancer and its precursors.” Future Oncol. 2010, 6, 7: 1143–1154. doi:10.2217/fon.10.79
[5] De Veld DC, Witjes MJ, Sterenborg HJ, Roodenburg JL. “The status of in vivo autofluorescence spectroscopy and imaging for oral oncology.” Oral Oncol. 2005, 41: 117–131.
[6] Roblyer D, Richards-Kortum R, Sokolov K, et al. “Multispectral optical imaging device for in vivo detection of oral neoplasia.” J Biomed Opt. 2008, 13, 2: 024019.
[7] Becker-Hickl. Metabolic Imaging by NAD(P)H and FAD Film. Web: https://www.becker-hickl.com/applications/metabolic-imaging/. Accessed December 2019.
[8] Pu, Yang et al. “Changes of collagen and nicotinamide adenine dinucleotide in human cancerous and normal prostate tissues studied using native fluorescence spectroscopy with selective excitation wavelength.” Journal of Biomedical Optics 2010, 15, 4. Print.
[9] Pena, A.M. et al. “Spectroscopic analysis of keratin endogenous signal for skin multiphoton microscopy.” Abstract, Laboratory for Optics and Biosciences, CNRS/INSERM, Ecole Polytechnique: 2005.
[10] ThorLabs. “Fluorescent Microscope Slides and Alignment Disks.” Web: https://www.thorlabs.com/newgrouppage9.cfm?objectgroup_id=12142. Accessed December 2019.
[11] Joyce Farrell and Brian Wandell 2019, ISETCam, "https://github.com/ISET/isetcam".
Appendix I
ISETCam ISETFluorescence was used to analyze measurements from the OralEye Camera [11].
Appendix II
Anand and Persiana worked on characterizing the fluorophores by measuring the absorption and emission spectra of each fluorophore. They used a fluorescence spectrometer (fluorimeter) in the chemistry department that can measure wavelengths from the UV range to 1,000 nanometers as well as has a sub-nanometer image resolution. Following fluorimeter training, preparation of samples, and recording of measurements, Anand and Persiana analyzed the data and interpreted it for the writing of this report and the presentation.
Based on the results of the fluorophore absorption and emission spectra, Sofie and Ramya constructed a matrix containing the optimal water-soluble fluorophores. The design was modeled after that of Connect Four, and was 3D-printed on campus, in the Huang basement. Following construction of the matrix, Sofie and Ramya proceeded to take measurements with the OralEye Camera in Dr. Farrell’s lab. They then subsequently plotted their results using MATLAB scripts found in Appendix I.
We all contributed to the final in-class presentation, and Sofie, Persiana, and Ramya wrote up our work for the Wiki page submission.
Acknowledgements
We would like to express our appreciation for the countless hours that Professor Brian A. Wandell, Dr. Joyce E. Farrell, and Zheng Lyu spent on making this course truly exceptional. Across our team, the class has inspired several new interests, including presenting magneto-sensor research at the next imaging conference, exploring a career in ophthalmology, and continuing to refine our fluorophore results in the year to come. We are tremendously grateful for your time, energy, and investment in our team. It brought us great joy and honor to be your students.
